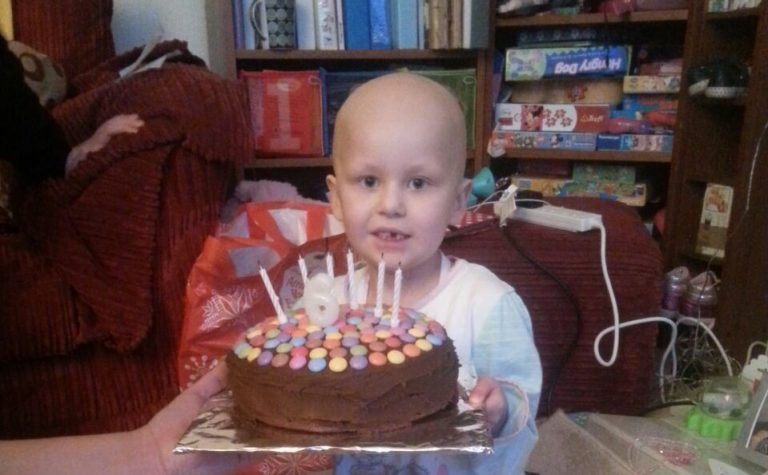
The pilot including more than 200 children found that half had gene mutations that are targetable by adult cancer drugs that are either available as standard treatment or via clinical trials.
- NHS pilot found half of children had mutations in their tumours that would be targetable by drugs available for adults
- Those children who did receive targeted treatments saw significant benefits
- But only 7 per cent of children with ‘clinically actionable’ mutations could access appropriate drugs, partly due to regulatory and funding barriers, and lack of access to clinical trials
- The pattern of mutations in tumours changed after standard treatment – showing importance of tracking cancer evolution over time
Although few children on the study went on to receive adult drugs, those who did receive targeted therapies had significant benefits. But the study also revealed the regulatory and funding barriers to children receiving the newest drugs, as only 7 per cent of those with targetable mutations were able to access the appropriate adult drug. The study was led by The Institute of Cancer Research (ICR), London, and The Royal Marsden NHS Foundation Trust, and offered genetic testing of tumours to children as part of a clinical trial. Some 20 additional hospitals around the UK participated by sending children’s biopsies in for testing. Researchers used a gene panel test to read the DNA sequence of 91 genes that drive cancer’s growth and spread from 223 children’s tumour biopsies – looking for potentially ‘targetable’ mutations. Solid tumours – such as those of the brain, central nervous system, bone and muscle – are rare but have much worse survival rates than children’s blood cancers such as leukaemia. Surgery is often not possible and treatment is limited to ‘blunt instrument’
chemotherapies. Using the panel test, researchers found 51 per cent of tumour samples tested had mutations that could be targeted by adult cancer drugs. For example, several children had BRAF gene mutations – which are common in melanoma skin cancers and can be treated using melanoma drugs. Using these drugs, one of the children had their
brain tumour held in check for 13 months before developing resistance. Another was on the drug for nine months with no progression of disease. But there are still challenges to overcome, since the majority of children with targetable mutations didn’t receive adult drugs – because there was no trial available for the drug in children, they were unable to access the drug on the NHS or they were too ill to receive an experimental treatment by the time they were tested. For 12 of the children, the researchers were also able to test for cancer gene mutations in DNA released from tumours into the bloodstream from a blood sample. They found blood tests picked up almost all of the mutations found in the tumour, and in some cases they also found extra mutations which were not detected in the tumour region biopsied. In future work the researchers will use serial blood tests to monitor how tumours evolve in response to therapies – which will be particularly useful in hard-to-biopsy tumours. Additionally, for children with brain tumours, the researchers are now looking at using samples of cerebral-spinal fluid to find drug targets. Although lumbar punctures are invasive, they are less so than a brain biopsy. Study author Dr Sally George, Clinical Research Fellow at the ICR, said:
Children deserve the very best cancer treatments, so they can live as long as possible and as well as possible. We desperately need better, more intelligently designed treatments which can give children longer with their families with fewer side effects. By testing tumours for specific gene mutations, we have shown it’s possible to identify new smarter, kinder treatment options for children, which may potentially give these patients much longer with their families after conventional therapies have failed. But our study also exposes the desperately frustrating barriers that children still face in receiving new treatments – barriers which lie in the regulations controlling how drugs for children are developed and approved.
Study leader Professor Louis Chesler, Professor of Paediatric Cancer Biology at the ICR, said:
In future, I want to be able to treat more children whose tumours have these targetable mutations with better drugs, as currently not all children have access. Gathering the molecular data will be good evidence to more clearly guide use of the most appropriate drug for each child. It is also very important that we extend robust and detailed testing to children at time of diagnosis, so we can more accurately classify and treat these cancers in the first place.
Dr Mike Hubank, Head of Clinical Genomics at The Royal Marsden NHS Foundation Trust, said:
The next steps for testing will be to look at using liquid biopsies to detect targetable tumour mutations without having to rely on invasive biopsies to get the information. Our early results, presented here, show that we can detect more mutations in blood than we do in conventional biopsies. It is probably in the blood that we get a more complete picture of the whole tumour, and not just the small part of the tumour that was removed for testing. Blood-based testing will also allow us to monitor tumour response to treatment and may be able to detect relapses early, offering the possibility of finely tuned, personalised treatments in the future.
Karen Capel, founder of charity Christopher’s Smile, who lost her son Christopher to
medulloblastoma in 2008, said:
When our son died there was no biological information available to doctors about individual children’s tumours. There is an urgent unmet need to provide new treatments for those children diagnosed with the most aggressive and hard-to-treat cancers. This test Professor Chesler and colleagues at the ICR developed is a first for children. We believe gene sequencing is the key foundation stone in enabling personalised medicine, and it will help to bring new treatments for children a step closer. Building on the foundations of the sequencing test, blood tests could provide critical information for any child from diagnosis throughout their treatment and into remission – opening the door for additional, continued or changed treatments. We are determined to fight for these liquid biopsies to become standard of care at the earliest opportunity.
The research was published in the European Journal of Cancer and was primarily funded by Christopher’s Smile and the NIHR Biomedical Research Centre at The Royal Marsden NHS Foundation Trust and the ICR.